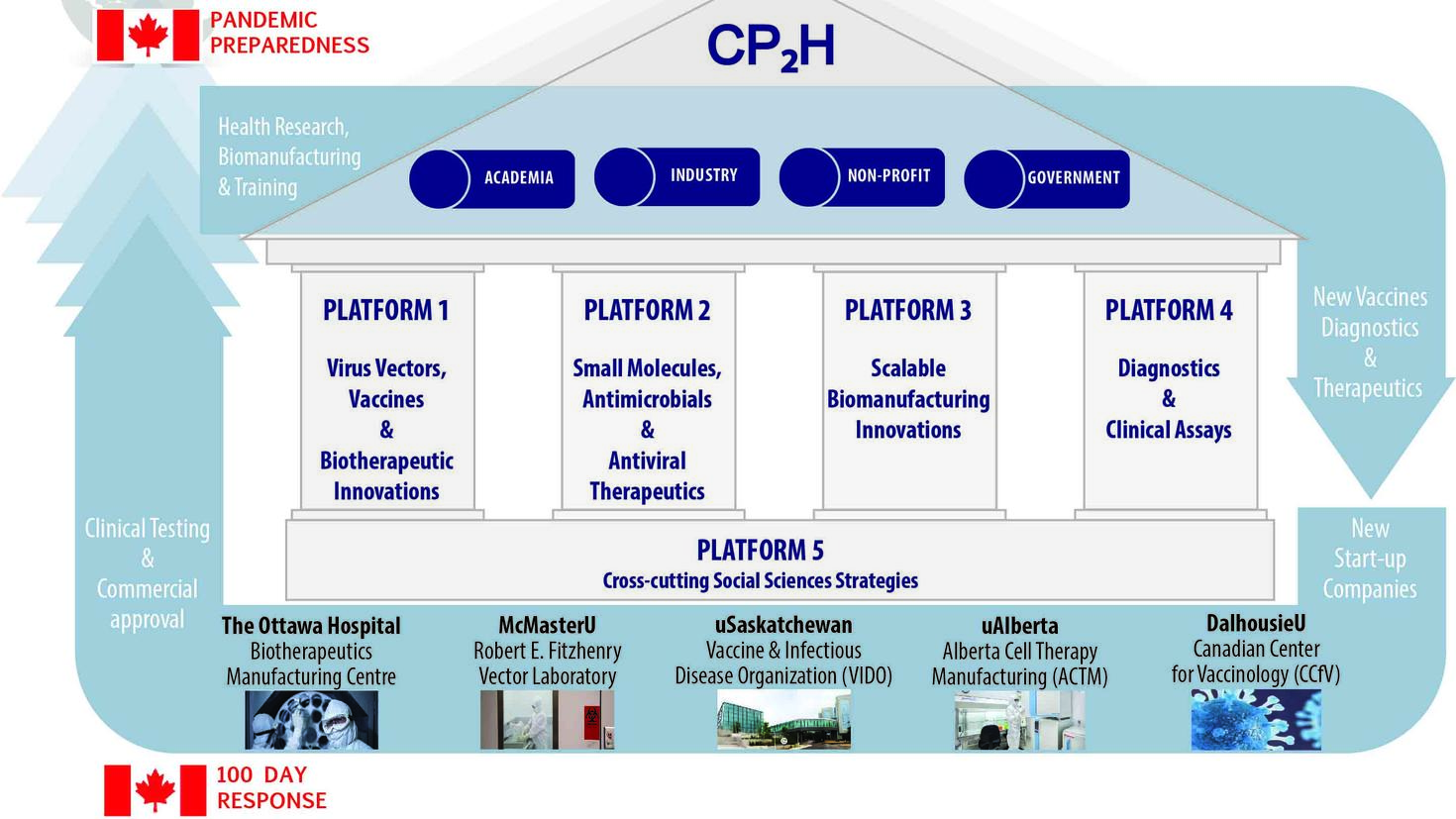
The Coalition for Epidemic Preparedness Innovations (CEPI) has set forth an ambitious 100-day challenge for G7 countries to make unprecedented investments in diagnostics, vaccine and therapeutic R&D, biomanufacturing, clinical development and regulatory changes to meet this challenge.
The Canadian government response has been a $2.1 billion investment in Canada’s Biomanufacturing and Life Sciences Strategy that supports Stage 1 funding for the five Canada Biomedical Research Fund (CBRF) hubs (including CP2H) and the launch of the CBRF and Biosciences Research Infrastructure Fund Stage 2 Competition supported by the CBRF hubs.